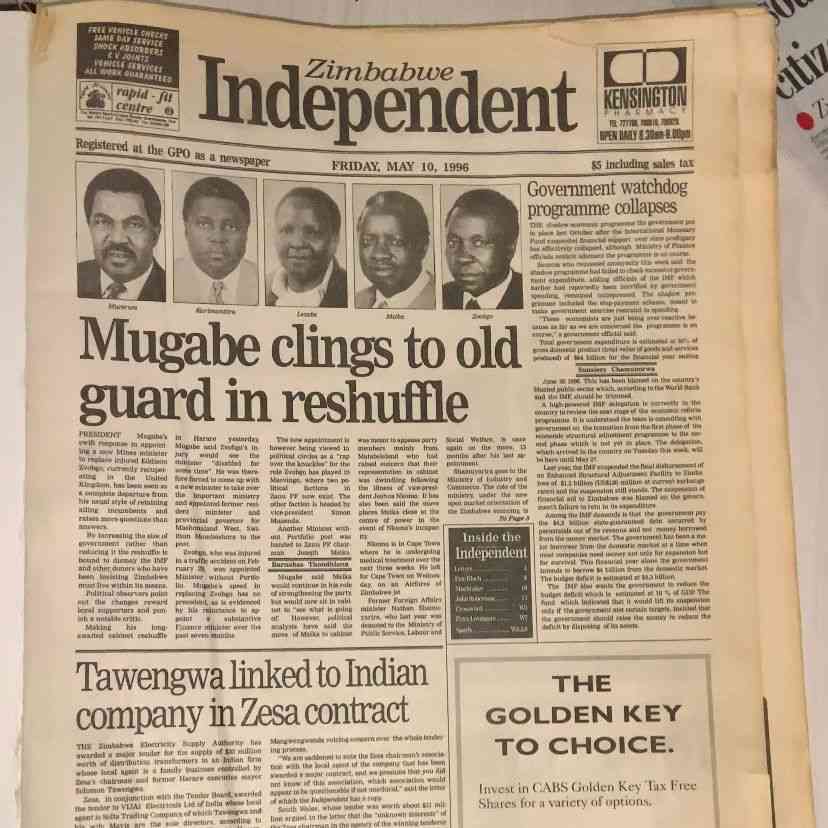
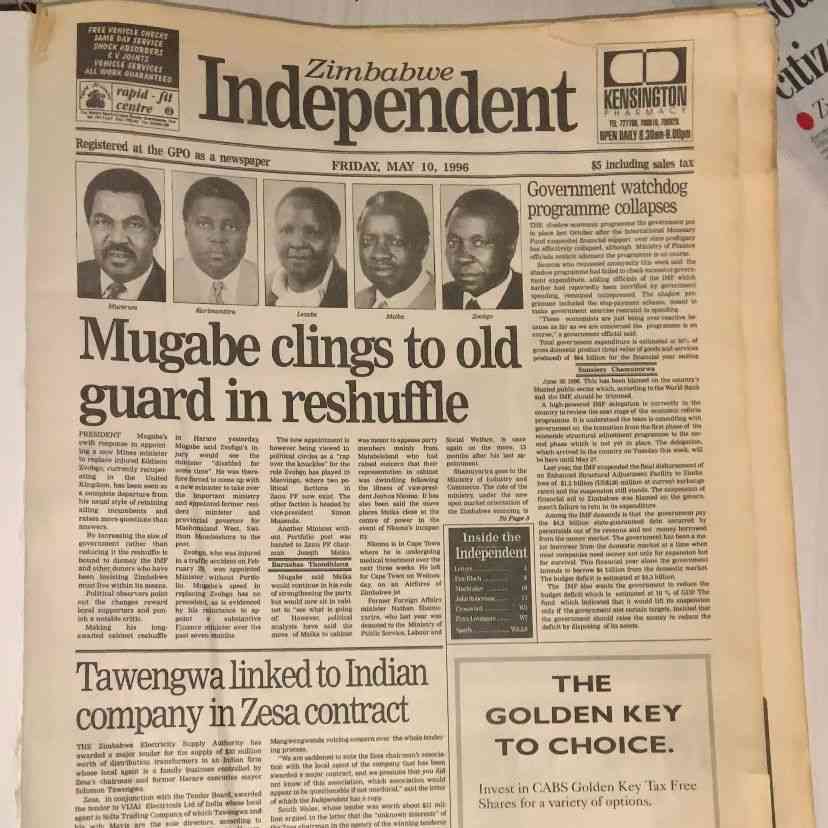
ZIMBABWE’S pharmaceutical manufacturing industry requires about US$45 million in fresh capital to upgrade production facilities and meet international quality standards, sector leaders said this week, arguing such investment could unlock access to a Southern African medicines market worth more than US$3 billion annually.
Despite operating at roughly 50% capacity utilisation, local manufacturers continue to face structural constraints that limit their ability to compete regionally.
Zimbabwe imports between 60% and 70% of its medicinal requirements, underscoring both domestic supply gaps and the scale of the opportunity.
Pharmaceutical Manufacturers Association of Zimbabwe chairperson Shepherd Mudzingwa said targeted capital injection was essential if the country is to reposition itself as a competitive supplier within the region.
“To make Zimbabwe’s pharmaceutical industry regionally competitive, the sector needs an estimated US$45 million to fund plant upgrades to at least World Health Organisation Good Manufacturing Practice benchmarks,” Mudzingwa said.
Achieving World Health Organisation (WHO) Good Manufacturing Practice (GMP) certification is widely regarded as a gateway to export markets, as it assures regulators and buyers that medicines are consistently produced and controlled according to international quality standards.
Mudzingwa said raising production standards would enable local firms to tap into new markets across southern Africa and beyond.
“The Southern African Development Community imports more than US$3 billion worth of pharmaceuticals each year. Many of these products could potentially be manufactured within the region.”
- Little hope for Zim, Africa
- African leaders dying overseas expose cruel deception of independence
- Sadc must intervene on Zim crisis: SA
- Sadc meets over water, energy and food security
Keep Reading
Zimbabwe’s pharmaceutical industry has long been identified as a strategic pillar for strengthening national health security and advancing industrialisation.
However, limited reinvestment, ageing equipment and heavy reliance on imported active pharmaceutical ingredients have slowed expansion.
Industry data shows that Zimbabwean manufacturers produce a range of generic medicines, including antibiotics, analgesics and chronic disease treatments. Yet domestic output remains insufficient to meet local demand, forcing the country to source most medicines from India, China and parts of Europe.
Regionally, the picture is similar. Across the Southern African Development Community (Sadc), pharmaceutical production remains concentrated in a few economies, notably South Africa, while many member states rely heavily on imports. The Covid-19 pandemic exposed vulnerabilities in global supply chains, prompting renewed calls for regional value chain development and greater self-sufficiency in essential medicines.
Under the African Continental Free Trade Area framework, there is growing emphasis on building regional manufacturing hubs capable of serving multiple markets.